DNP Project IRB Approval Tips

Before initiating a Doctor of Nursing Practice (DNP) project involving human participants, one must obtain approval for the DNP project from an ethics review committee, typically referred to as the Institutional Review Board (IRB). Upon receiving IRB applications for DNP projects, committee members are responsible for (i) conducting an initial review of studies with participants, (ii) handling issues related to research involving human subjects, (iii) approving the waiver of informed consent, and (iv) endorsing the research with human participants. In case you are a nursing student in search of IRB approval help for DNP programs to ensure that your project meets ethical and institutional standards, our professional nursing writers can help. Our professional writers are familiar with the Belmont Report principles and clearly understand the federal guidelines for IRB application.
In addition, our IRB application experts have over a decade of expertise, making them acquainted with the different types of IRB reviews and know the common reviewers’ concerns and how to address them. We specialize in areas such as IRB form filling, revising application forms based on requested changes, and IRB review type identification, among others. In this article, we have discussed some essential tips that DNP students can apply to achieve smooth approval to conduct their projects, such as ensuring respondents are properly informed, submitting all required IRB forms and supporting documents, and protecting participants’ confidentiality.
What is DNP Project IRB Approval?
The IRB aims to protect participants from psychological or physical harm by reviewing research protocols of the DNP project to determine the ethics of the research, promote voluntary participation by respondents, and ensure the safety of the individuals. DNP project IRB approval is a practice that involves IRB committee members reviewing and accepting the carrying out of an advanced nursing project with human subjects to ensure the ethical and legal protection of participants. By evaluating and approving the DNP project research, the IRB committee ensures that the ethical guidelines are followed to protect the welfare of respondents as scholars contribute to evidence-based research.
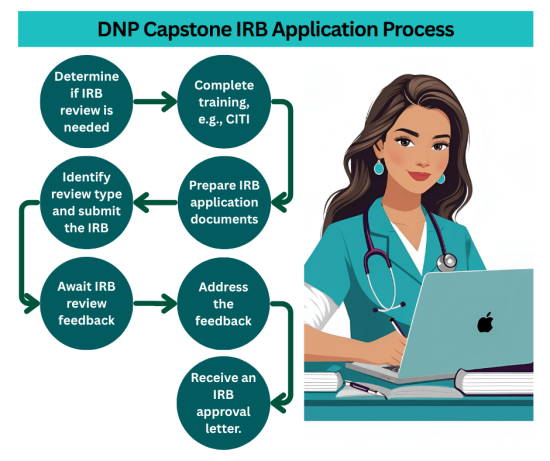
Types of DNP Projects That Require IRB Approval
DNP projects engaged in human participant research must get IRB approval before collecting and analyzing data for the study. DNP projects that require IRB approval include some quality improvement projects that involve human respondents, program development, and clinical practice guidelines studies. IRB approval is essential for ensuring that the DNP project is ethical, aligns with federal research and university guidelines, and protects participants’ rights.
Expert Tips to Speed Up IRB Approval for DNP Projects
1. Ensure Completion of Necessary CITI Trainings Before Application
The Collaborative Institutional Training Initiative (CITI) is a provider of web-based research education information that aims to facilitate the training of investigators and all project team members. The CITI training program aims to improve the integrity and professionalism of medical students conducting research and other compliance-related activities, and promote ethical research through the education of nursing students. Before applying for IRB approval, the nursing student should ensure every member involved in the DNP project has received formal ethics training, which is essential for the IRB application process and protection of investigators and human subjects conducting the study. Through CITI training before IRB application for the DNP project, scholars are able to establish credibility of their study, learn the best ethical practices, and comply with set regulations in their nursing programs.
2. Clearly Explain the Research Plan of the DNP Project
A research plan is a documented overview of the entire DNP project from the beginning of the research to the results expected at the end of the study. When seeking IRB approval for the DNP project, the nursing scholar should develop a comprehensive research plan that consists of:
- Introduction and background.
- Specific aims and objectives.
- Methods, materials, and analysis.
- Research population, recruitment methods, and compensation.
- Informed consent process.
- Participant privacy, data disposition, and data confidentiality.
- Potential risks or discomfort to participants.
- Benefits of the DNP project.
- DNP students’ qualifications, roles, and training.
Composing a detailed research plan for the DNP project enables the IRB to determine if the approval criteria for human participants’ research, as illustrated in the federal regulations, are met.
3. Ensure Respondents are Adequately Informed
Informed consent is the provision of detailed information to potential respondents to be involved in the DNP project, enabling them to make a decision about their participation. To ensure that the human subjects are well informed, the DNP scholar should disclose any necessary information, facilitate understanding, and promote voluntary decision-making. The nursing student should utilize plain language in the consent documents in layman’s terms to accommodate various participant populations. By presenting clear consent procedures, the DNP student demonstrates adherence to ethical standards and protection of participants, therefore facilitating approval by the IRB.
4. Submit All Required IRB Forms and Supporting Documents
The IRB reviews forms and supporting documents for the application to determine if the DNP project meets the criteria for IRB approval of research and other federal regulations. Some examples of forms and documents that the DNP scholar should submit for IRB approval include, but are not limited to, the IRB review, CITI certificate, research and recruitment protocols, informed consent form, and support letters. Submitting all required papers for the IRB application ensures ethical research practices and compliance with set regulations, therefore enabling quick approval.
5. Identify and Mitigate Potential Risks
Two of the required criteria for granting IRB approval for research are that risks to subjects are minimized by utilizing a sound research design that does not expose participants to harm, and potential threats to respondents are reasonable in relation to anticipated benefits. As a DNP student, participant risks can be avoided in the DNP project by involving expert investigators, collecting data from standard of care procedures, incorporating safeguards into the study design, and elaborating on the precautions to be applied to the population of interest. By documenting any potential risks, how they will be minimized, and the DNP student’s qualifications and plans to protect the rights of the participants involved in the nursing project, the IRB submission is more likely to be approved.
6. Clearly Explain Data Usage, Analysis, and Storage Plans
The measure of quality of the DNP project is determined by how the scholar has described the data analysis process and data storage procedures and how they have applied ethical requirements to their project. The IRB review committee usually evaluates whether the sample size is large enough to provide sufficient data, a description of specific variables, analysis techniques, and how the nursing scholar will determine if objectives have been met. Providing a detailed description of how data will be utilized, analyzed, and stored ensures the validity of the DNP project, therefore increasing the chances of IRB approval for implementation.

What Mistakes are Most Likely to Affect the DNP Project IRB Approval?
- The DNP project faculty advisor did not sign the certification and approval section.
- The informed consent form does not include all the required sections or has inconsistent information in the application.
- Any DNP project risks are not illustrated, even when very minimal.
- If the confidentiality of data is not addressed adequately.
- Lack of essential documentation, such as the questionnaire or surveys.
- Methods of participant recruitment are not adequately described.
- Documents have grammatical or structural errors and are hard to follow.
Summary
The IRB aims to protect respondents from potential risks by reviewing the research protocols of research involving human subjects. Thoughtful consideration of research participants through the IRB application process demonstrates the DNP scholar’s adherence to evaluation and ethical standards. Following the suggested tips is essential for strengthening nursing scholars’ applications, complying with federal regulations, and increasing the likelihood of timely approval.
Following the suggested tips is essential for strengthening nursing scholars’ applications, complying with federal regulations, and increasing the likelihood of timely approval. In case you need professional help with an IRB application for the DNP project, contact us or chat with our knowledgeable and friendly customer service team. We have a proven track record of successful IRB approvals; hence, no need to worry, just leave us your request for an IRB application for a DNP project.
